
Tirzepatide Deep Dive: Dosing, Results, and What Patients Should Expect
Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
Trying to keep up with the news on Testosterone Replacement Therapy (TRT) can feel like getting whiplash. One minute you hear about potential risks, the next you see headlines saying it’s a vital part of men’s health. It’s confusing, especially if you’re on TRT or just wondering if it’s right for you.
Well, things just got a whole lot clearer. In late February 2025, the U.S. Food and Drug Administration (FDA) updated its official guidelines on testosterone products, and it’s a pretty big deal. These changes weren’t random; they came from some serious, long-term clinical research.
So, what’s the real story? We’re here to cut through the noise. This article will give you a straightforward, fact-based look at what changed, why it changed, and what top experts are saying about where TRT is headed. By the end, you’ll have a much better handle on making smart decisions about your health in 2026.

The FDA didn’t just wake up one day and decide to change its mind on TRT. The decision was a direct result of a massive clinical trial designed to answer a question that’s been debated for years: is it safe for your heart?
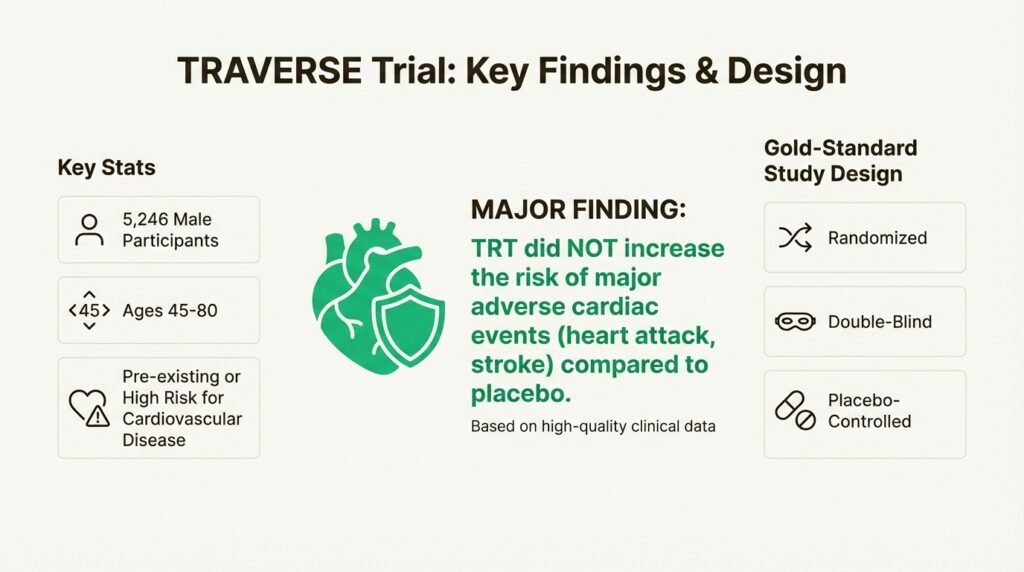
For a long time, the medical community has been dealing with conflicting reports about TRT and heart health. To settle things once and for all, a huge study was launched, called the Testosterone Replacement Therapy for Assessment of Long-term Vascular Events and Efficacy Response in Hypogonadal Men (or just TRAVERSE).
This wasn’t a small-scale study. The TRAVERSE trial involved 5,246 men between 45 and 80, all of whom already had cardiovascular disease or were at high risk for it. It was a randomized, double-blind, placebo-controlled study, which is the gold standard for research. The main finding was huge: the trial showed that for these men, TRT was “noninferior to placebo with respect to the incidence of major adverse cardiac events” like a heart attack or stroke. Basically, it didn’t increase the risk. This high-quality data finally gave the FDA the solid evidence it needed to clear up years of uncertainty.
To get the full picture, you have to rewind a bit. The FDA’s cautious position started back in 2014 when they issued a Drug Safety Communication because of those conflicting reports about heart risks.
This led to a major decision in March 2015, when the FDA required all testosterone manufacturers to add warnings about a possible increased risk of heart attack and stroke to their labels. More importantly, they ordered these companies to conduct a large, well-designed clinical trial to get to the bottom of the cardiovascular risk. The TRAVERSE trial was the result of that order, and its findings provided the strong evidence everyone had been waiting for.
It’s important to know the difference between what the FDA has officially changed on product labels and what an expert panel has recommended for the future. This section covers the official updates that are in effect right now.

This is the big one. In late February 2025, the FDA announced it was officially removing the Boxed Warning language that warned of an increased risk of heart attack and stroke.
This change was a direct response to the clear findings from the TRAVERSE trial. After years of caution, the best available science showed that the risk wasn’t what some earlier, smaller studies had hinted at. For men on medically supervised TRT, this is a huge relief.
While the heart attack risk warning is gone, something new has taken its place. The FDA has added a warning about a class-wide increase in blood pressure linked to testosterone use. This decision was based on a different set of studies, called ambulatory blood pressure monitoring (ABPM) studies, which the FDA has required since 2018.
This new warning doesn’t mean TRT is unsafe; it just means one of its potential side effects needs to be watched closely. This is exactly why professional, hands-on monitoring is so important. Managing potential side effects like blood pressure is a key part of the comprehensive care you’d get at a wellness center like Body Works, where the goal is to make sure your treatment is both safe and effective. It’s not just about getting a prescription; it’s about having a team that looks at your whole health picture.
One thing that hasn’t changed is the FDA’s official position on who should be prescribed testosterone. The approved use is still for men with low testosterone levels caused by specific medical conditions, like genetic disorders or damage from chemotherapy. This is often called “classic hypogonadism.”
Officially, TRT is not approved for treating low testosterone caused only by aging. This has been a major point of debate for years and is exactly what the expert panel, which we’ll cover next, wants to see changed.
While the FDA’s official label changes are a big step, a meeting of top experts in December 2025 suggested that even bigger shifts could be coming. This FDA expert panel, full of leading urologists and health officials, argued for changes that go way beyond the recent updates, hinting at a fundamental shift in how the medical community sees TRT.
The panel made several bold recommendations that could reshape how TRT is accessed and viewed in the coming years.
Broader eligibility: The experts argued for expanding TRT access to men who have symptoms of low T and confirmed low levels, even without a specific underlying medical condition like a pituitary tumor. This approach is in line with modern guidelines from groups like the American Urological Association (AUA).
Removing “controlled substance” status: One of the biggest suggestions was to reclassify testosterone from a Schedule III drug. They argued that its current status creates unnecessary stigma and paperwork, making it harder for primary care doctors to prescribe it to men who need it.
Routine screening: Panelist Dr. Helen Bernie, an Associate Professor of Urology, argued that low testosterone should be seen as a “critical biomarker” for a man’s overall health and should be screened for regularly, just like high cholesterol or diabetes.
The driving force behind these recommendations was a shared belief that the medical community is finally moving past old, outdated fears about TRT. Panelist Dr. Mohit Khera put it powerfully, stating that there have been “decades of misconceptions” about TRT’s risks, especially around prostate cancer and heart disease, that modern data has largely debunked.
The panel’s message was clear: the current restrictive labeling is “not fit for purpose” anymore. It’s stopping countless men who could benefit from therapy from getting the treatment they need.
“Testosterone therapy is not a lifestyle drug. It is a cornerstone for preventive health.”
– Dr. Helen Bernie, Associate Professor of Urology, Indiana University (FDA Expert Panel, Dec. 2025)
With all these changes, the TRT landscape is looking more favorable and evidence-based than ever. But this doesn’t mean it’s a free-for-all. If anything, it makes expert medical supervision even more important. Here’s how you can handle it all safely.
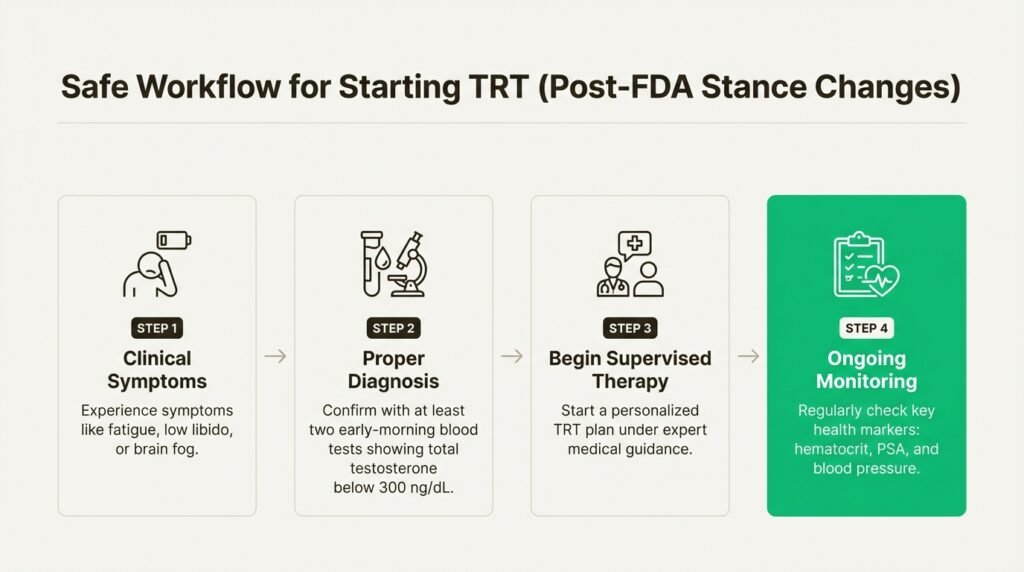
Let’s get one thing straight: TRT is not a DIY project or something you should guess at from an online quiz. According to AUA guidelines, a proper diagnosis of low testosterone requires two things: clinical symptoms (like fatigue, low libido, or brain fog) and at least two early-morning blood tests showing your total testosterone is below 300 ng/dL.
Once you’re on therapy, the work isn’t over. Ongoing monitoring is crucial to make sure the treatment is working correctly and safely. This means regular checks of key health markers like your hematocrit (red blood cell concentration), PSA (for prostate health), and, given the new FDA warning, your blood pressure. This is what a responsible, medically supervised program looks like.
It’s really important to get the difference between medical TRT and using testosterone for bodybuilding or performance enhancement. The two are worlds apart.
Medical TRT is all about replacement. The goal is to bring low hormone levels back into a healthy, normal range. The AUA suggests aiming for a level in the middle of the normal range, typically around 450-600 ng/dL. This is about restoring balance and improving your quality of life.
On the other hand, using testosterone for performance enhancement often involves taking massive doses, far beyond what the body would ever produce naturally. This approach carries much higher risks and isn’t based on sound medical practice. This is where expert guidance is so valuable. At a wellness center like Body Works, the entire focus is on health optimization under strict medical protocols. It’s about helping you feel your best safely, not pushing your body beyond its natural limits.

When you add it all up – the symptoms, the diagnosis, the right dosage, managing side effects, and keeping up with changing rules – it’s a lot to handle. This isn’t something you should try to figure out on your own.
A specialist can create a treatment plan tailored to your specific health profile, symptoms, and goals. They make sure you get the benefits of therapy while actively managing any potential risks, like the newly highlighted increase in blood pressure. This personalized, hands-on approach is the key to doing TRT the right way.
The world of TRT is changing for the better. The FDA has officially updated its stance, getting rid of the old cardiovascular boxed warning based on solid evidence from the TRAVERSE trial, while also adding a new warning about blood pressure to keep patients safe. At the same time, a panel of the nation’s top experts is pushing for even wider access, signaling a major shift in how the medical community views testosterone’s role in men’s health.
What this all means is that in 2026, it’s becoming safer and more supportive for men who have a real medical need for TRT. But this progress also highlights one non-negotiable fact: a responsible, medically supervised approach is absolutely essential.
If you’re experiencing symptoms of low testosterone and are wondering how these FDA TRT stance changes might affect you, the smartest first step is to talk to a professional. Contact Body Works at our Franklin or Nolensville clinic to schedule a comprehensive evaluation and create a personalized health plan that’s right for you.
Medically reviewed by Dr. Donald Vollmer, MD
Managing Physician, Body Works TN

Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
The weight gain that arrives during perimenopause is genuinely different from weight gain at other times of life. It often shows up around the midsection

For most of 2023 and 2024, semaglutide was on the FDA shortage list. That single regulatory designation opened the door for compounding pharmacies to legally

The most common question patients ask after their first injection is some version of “when will I see results?” That question is reasonable, but the
Hair shedding ranks consistently among the top concerns patients raise before starting semaglutide or tirzepatide. Search volume on “does semaglutide cause hair loss” has climbed
Most patients on semaglutide or tirzepatide eventually ask the same two questions: how long do I need to take this, and what happens if I