
Tirzepatide Deep Dive: Dosing, Results, and What Patients Should Expect
Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
The most common question patients ask after their first injection is some version of “when will I see results?” That question is reasonable, but the answer is not a single number. Semaglutide produces a predictable pattern of changes that unfolds over months, with appetite shifts visible in the first week, scale changes accelerating around weeks four to eight, and the largest single drops happening between months three and twelve. Knowing what to expect at each phase prevents two common mistakes: getting discouraged early when the scale moves slowly, and getting impatient mid-treatment when the rate of loss naturally slows.
This is a week-by-week and month-by-month guide to what the clinical evidence shows, what most patients actually experience, and how the dose titration schedule shapes the curve. The numbers come from the STEP 1 trial of semaglutide 2.4 mg, which is the most rigorous long-term data available, with real-world variation noted where it matters.
If you are still deciding whether semaglutide is right for you, the broader question of candidacy may be a better starting point. This guide assumes you already know semaglutide is a clinical fit and you want to understand the trajectory once treatment begins.

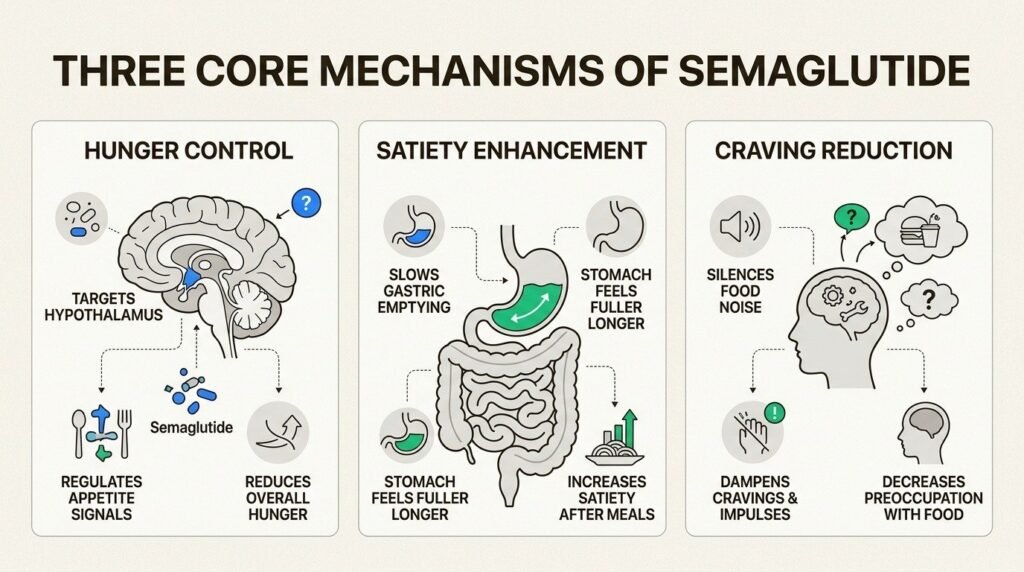
Before walking through the timeline, a quick note on what the medication is doing. Semaglutide is a GLP-1 receptor agonist. It mimics a gut hormone that signals the brain when you have eaten enough. The result is two complementary effects: meals satisfy you sooner, and the constant background pull toward food, what patients describe as food noise, fades.
Semaglutide also slows gastric emptying. Food sits in the stomach longer, fullness lasts longer, and the gap between meals stretches without effort. The combination is what produces the trajectory described below: appetite shifts come first, scale changes follow, and the largest cumulative loss arrives between months three and twelve.
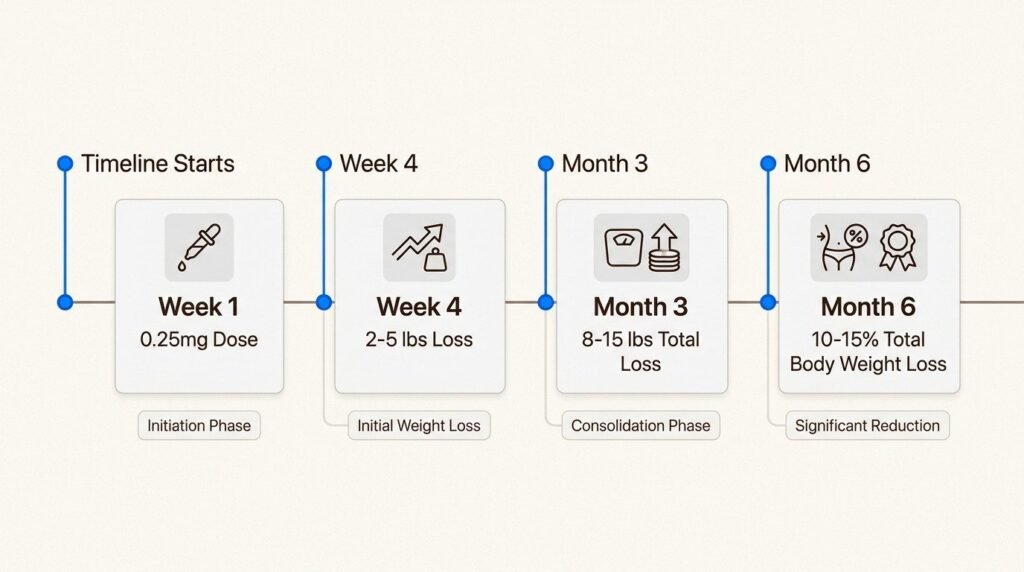
The first month is a titration phase, not a weight loss phase. You start at 0.25 mg once weekly. This dose is low enough that side effects are usually mild, but it is not high enough to drive substantial weight loss for most patients. The job of weeks one through four is letting your body acclimate.
What you should notice in the first week or two is a quiet but unmistakable shift in appetite. Meals feel like they finish sooner. Snacks between meals lose their pull. The constant low-volume thinking about food that many patients describe as food noise begins to fade. Mild nausea, especially in the first one to three days after each dose, is common and usually manageable with smaller meals and avoiding high-fat foods.
Scale changes during this phase average two to four pounds for most patients. Some lose more, some lose nothing, and both are within normal range. The 0.25 mg dose is doing its job if you are tolerating it and noticing appetite changes, even if the scale has barely moved.
At week five, the dose typically doubles to 0.5 mg weekly. Appetite suppression deepens. The fullness signal after meals lasts longer. Patients often report that they finish dinner without finishing the plate for the first time in years. Cravings for highly processed or sweet foods often diminish further.
Weight loss begins to accelerate during this window. Cumulative loss by week eight is typically four to eight pounds, or roughly two to four percent of starting body weight. Mild side effects, especially the day after injection, are still possible but usually less intense than the initial titration step.
This is also the phase where patients begin to notice non-scale changes. Clothing fit shifts before the scale shows large drops because fat loss tends to come from the midsection first, where most patients carry the highest concentration. Waist circumference is a useful supplemental measurement during this phase.
The dose continues to step up at four-week intervals: 1 mg at week nine, then 1.7 mg at week thirteen for most patients. This is the first time the medication is at a clearly therapeutic level for weight loss, and the rate of loss reflects that. Patients typically lose two to four pounds per month during this window, with cumulative loss reaching ten to twenty pounds by week sixteen.
Side effect intensity may bump up briefly with each dose increase and then settle. Nausea, if it occurred earlier, often returns for a few days after each step before the body adjusts. Constipation becomes more common at higher doses and benefits from increased water intake and fiber.
The visible changes during this phase are substantial. Most patients have dropped a clothing size, have visible facial slimming, and start receiving comments from people who have not seen them recently. Energy and sleep often improve. Patients who started semaglutide for a metabolic indication, like prediabetes or borderline hypertension, often see lab markers begin to move.
The full therapeutic dose for chronic weight management is 2.4 mg weekly, reached around week seventeen for most patients. This is the dose used in STEP 1, and it is where the largest sustained appetite suppression happens.
By week twenty, cumulative loss for the average patient is in the range of fifteen to twenty-five pounds, roughly eight to ten percent of starting body weight. Some patients reach this milestone faster, some slower, and the variation is normal.
Patients who reach 2.4 mg with manageable side effects are positioned for the longest part of the weight loss curve, which spans the next several months at the maintenance dose. Patients who tolerated the titration but find 2.4 mg uncomfortable can sometimes hold at 1.7 mg as a slower but still effective dose. This is a clinical decision your provider makes based on tolerance and response.
The second half of the first year is when the largest cumulative weight loss happens for most patients. The dose is stable. The body has adapted to the medication. Lifestyle habits built during titration are paying off. Loss continues at a rate of two to five pounds per month for most patients, though the pace gradually slows as you approach a metabolic floor.
The STEP 1 trial measured outcomes at week 68, roughly sixteen months. By that point, the average semaglutide patient had lost 14.9% of their starting body weight, compared to 2.4% in the placebo group (Wilding et al., New England Journal of Medicine, 2021). For a 220-pound patient, 14.9% works out to roughly 33 pounds. Around one-third of patients lose 20% or more of their starting weight during this window.
This phase is also where the work shifts. Resistance training matters more, because preserving muscle becomes essential as fat loss continues. Protein targets stay high. Sleep, hydration, and stress management become the variables that separate strong responders from average ones (National Institute of Diabetes and Digestive and Kidney Diseases). Body composition tracking, not just scale weight, gives you a clearer picture of what the medication is doing.
By month twelve, most patients are within a few pounds of where they will land on semaglutide alone. The curve flattens. The body’s set-point regulation has caught up with the medication’s appetite suppression, and weekly weight changes become small. This is not a failure of the medication. It is the natural endpoint of a treatment that lowered the body’s defended weight to a new, lower equilibrium.
Patients still have meaningful options at this point. Some continue at 2.4 mg as a long-term maintenance plan. Some work with their provider to switch to tirzepatide, which is mechanistically distinct and sometimes produces additional loss in patients who have plateaued on semaglutide. Some begin a tapering plan toward a lower maintenance dose. The decision depends on the gap between current weight and goal weight, the response so far, and individual tolerance.
About fifteen percent of patients are clinical “non-responders” to semaglutide, defined as patients who have lost less than five percent of body weight by week twelve at the therapeutic dose. Non-response is rarely about willpower. It usually reflects genetic and metabolic differences in how an individual processes GLP-1 receptor activation.
For patients whose loss is slower than expected, several adjustments are worth considering with a provider. Switching to tirzepatide sometimes produces a stronger response because it activates both GIP and GLP-1 receptors. Reviewing thyroid function, insulin sensitivity, and cortisol can identify a metabolic obstacle that the medication alone is not addressing. Reviewing actual food intake, often surprising even to careful patients, sometimes uncovers an unintentional caloric ceiling that has not really been a deficit.
A flat scale at month two is not a failure. A flat scale at month four at the therapeutic dose, with appetite reduction and lifestyle in place, is a signal that the protocol needs adjustment. That is a conversation with your provider, not a moment to abandon treatment.
Medically reviewed by Dr. Donald Vollmer, MD
Managing Physician, Body Works TN

Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
The weight gain that arrives during perimenopause is genuinely different from weight gain at other times of life. It often shows up around the midsection

For most of 2023 and 2024, semaglutide was on the FDA shortage list. That single regulatory designation opened the door for compounding pharmacies to legally
Hair shedding ranks consistently among the top concerns patients raise before starting semaglutide or tirzepatide. Search volume on “does semaglutide cause hair loss” has climbed
Most patients on semaglutide or tirzepatide eventually ask the same two questions: how long do I need to take this, and what happens if I

If you have been struggling to lose weight despite trying diet after diet, you are not alone. For many people in Franklin, TN, Nolensville, TN,