
Who Is a Good Candidate for Semaglutide? A Complete Guide
If you have been struggling to lose weight despite trying diet after diet, you are not alone. For many people in Franklin, TN, Nolensville, TN,

If you are researching medical weight loss, two medication names keep coming up: semaglutide and tirzepatide. Both have changed what is possible with non-surgical weight loss, and there is now head-to-head clinical data showing how they stack up against each other. But they are not interchangeable, and the right choice depends on how each medication works, your medical history, and how your body responds during treatment.
At Body Works in Franklin, TN and Nolensville, TN, physicians prescribe both semaglutide and tirzepatide and tailor the selection to each patient. According to the National Institute of Diabetes and Digestive and Kidney Diseases, GLP-1 receptor agonists now produce 3 to 12% more weight loss than lifestyle intervention alone. This guide walks through the latest clinical evidence, including the 2025 SURMOUNT-5 head-to-head trial, so you can have an informed conversation with your provider.
Both medications belong to a class called incretin mimetics, but tirzepatide engages a second hormone pathway that semaglutide does not. Semaglutide is a pure GLP-1 receptor agonist: it mimics glucagon-like peptide-1, a hormone your gut releases after eating. It acts on the appetite center in your hypothalamus, slows gastric emptying so food stays in your stomach longer, stimulates insulin release when blood sugar is elevated, and suppresses glucagon.
Tirzepatide is a dual GLP-1 and GIP receptor agonist. It activates the same GLP-1 pathway semaglutide uses, but it also targets glucose-dependent insulinotropic polypeptide (GIP) receptors. Researchers believe this dual action enhances insulin sensitivity and fat metabolism beyond what single-pathway GLP-1 treatment achieves, and may offer additional benefits for bone formation and kidney function.
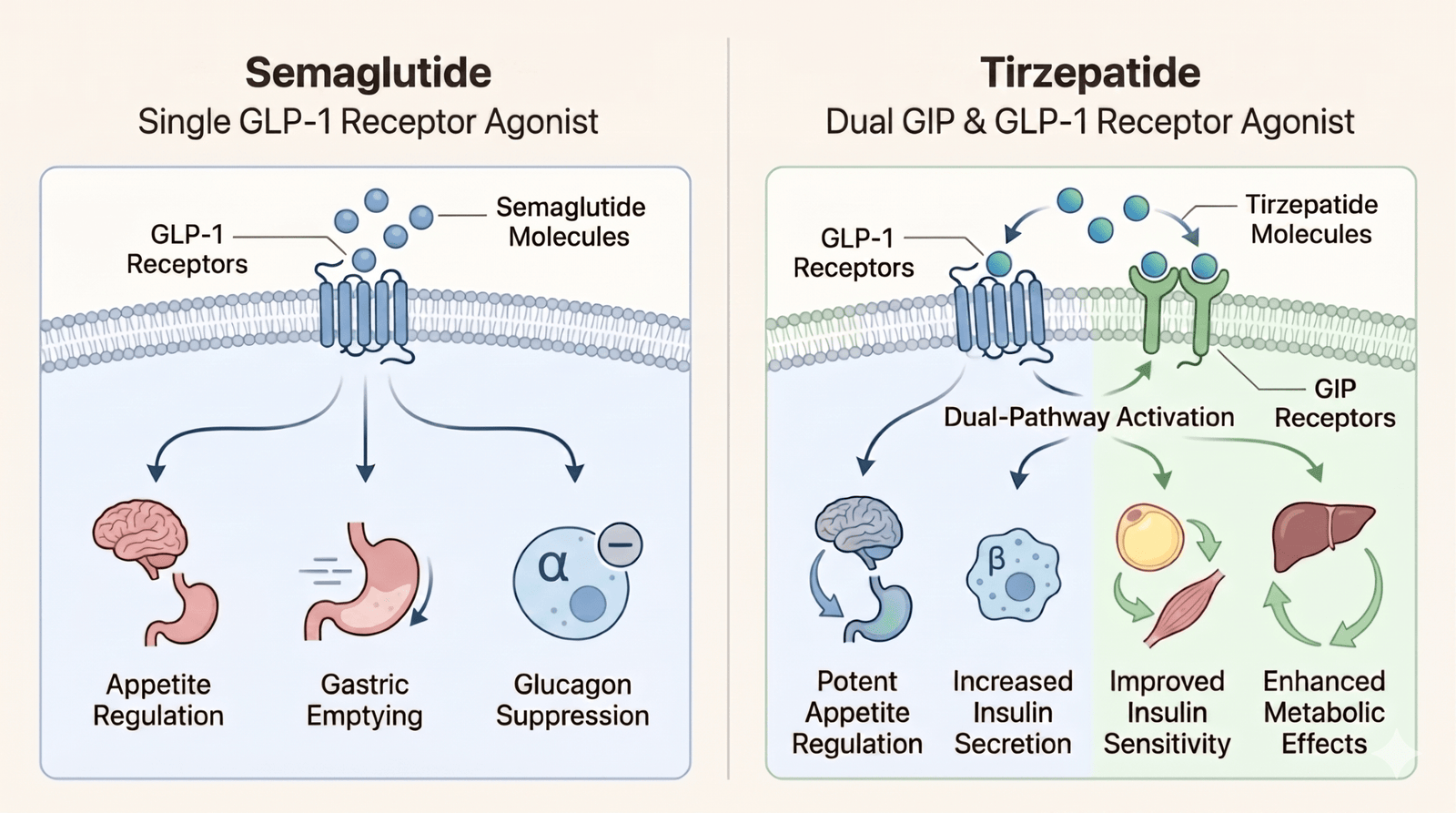
Both medications are taken as once-weekly subcutaneous injections and work best alongside a reduced-calorie diet and regular physical activity. If this drug class is new to you, our primer on what GLP-1 medications are and how they work covers the fundamentals before the comparison details below.
The SURMOUNT-5 trial, published in The New England Journal of Medicine in May 2025, is the first large randomized study to compare tirzepatide and semaglutide directly in adults with obesity. Over 72 weeks, tirzepatide produced notably greater weight loss across every metric the researchers measured.
| Outcome | Tirzepatide | Semaglutide | Difference |
|---|---|---|---|
| Mean body weight loss | -20.2% | -13.7% | -6.5 percentage points |
| Waist circumference reduction | -18.4 cm | -13.0 cm | -5.4 cm |
| Statistical significance | P<0.001 | P<0.001 | Highly significant |
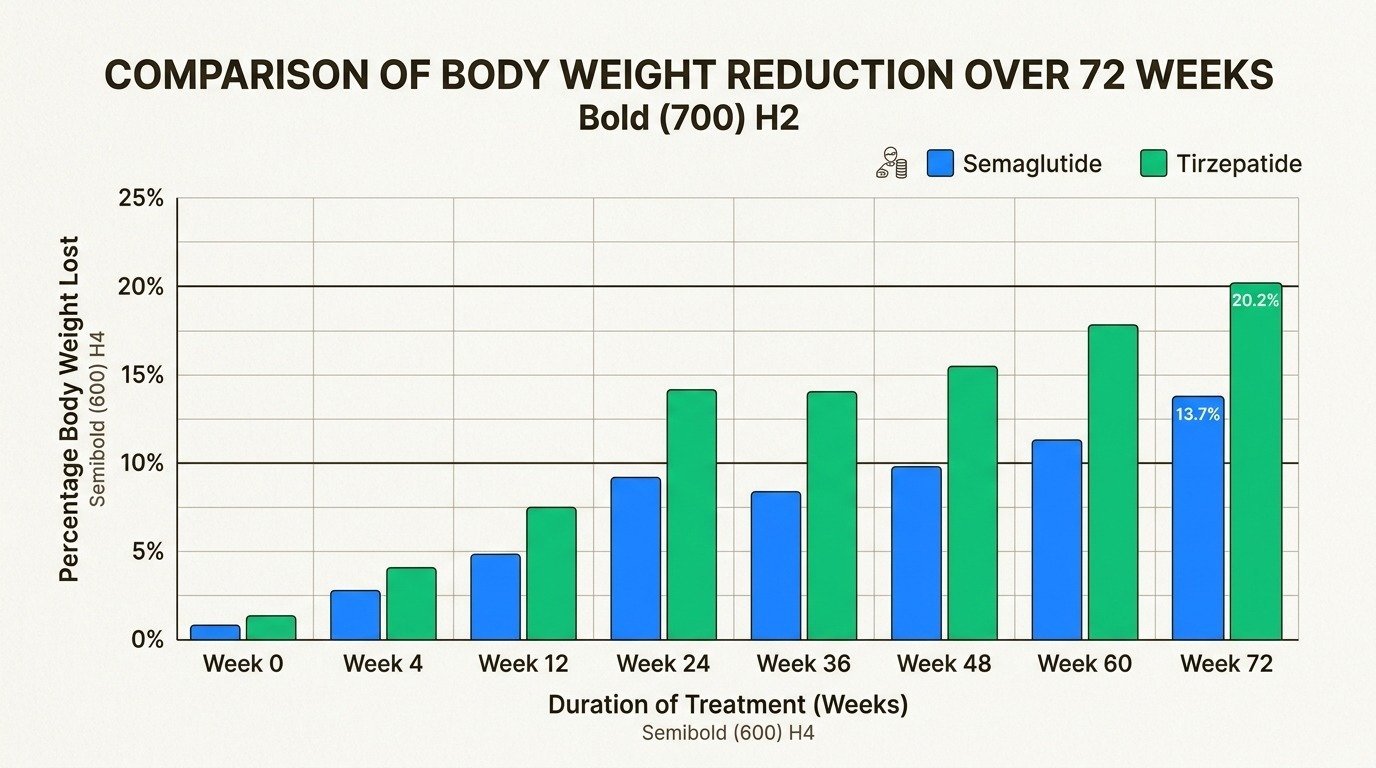
Significantly more tirzepatide patients hit every major weight loss milestone, including 10%, 15%, 20%, and 25% body weight reduction. Real-world data from electronic health records tells a similar story, though the gap narrows somewhat in patients who also have type 2 diabetes. These are averages, and individual results depend on starting weight, adherence to lifestyle changes, and metabolic factors. Our guide on how to succeed on medical weight loss injections walks through the habits that separate strong responders from weaker ones.
Both medications share the same gastrointestinal side effect profile, with nausea as the most common complaint. In the STEP 1 semaglutide trial (Wilding et al., NEJM, 2021) and the SURMOUNT-1 tirzepatide trial (Jastreboff et al., NEJM, 2022), nausea affected roughly 44% of semaglutide patients and about 24% of tirzepatide patients at the maximum dose. Diarrhea, vomiting, constipation, abdominal pain, and decreased appetite also occur in a meaningful minority of patients on both drugs.
Side effects are almost always worst during dose titration, when the medication is ramped up every 4 weeks. Most patients find symptoms fade as the body adapts. Gradual escalation over 16 to 20 weeks is the standard clinical protocol precisely because it minimizes GI symptoms while still reaching a therapeutic dose.
Both medications carry the same boxed warnings. They are contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN 2), and both carry warnings for pancreatitis, gallbladder disease, and acute kidney injury from dehydration. A 2024 safety review in ScienceDirect noted that tirzepatide may have slightly better GI tolerability than semaglutide, likely because of its dual mechanism (ScienceDirect, 2024).
Semaglutide currently has stronger evidence for heart health, making it the preferred option for patients with existing cardiovascular disease. The SELECT trial demonstrated that semaglutide 2.4 mg reduced major adverse cardiovascular events (heart attack, stroke, and cardiovascular death) by 20% in adults with overweight or obesity and established heart disease (Lincoff et al., SELECT trial, New England Journal of Medicine, 2023).
This was a landmark finding because it established that a weight loss medication could provide cardiovascular protection independent of diabetes status. Tirzepatide has not yet completed a comparable cardiovascular outcomes trial, though studies are underway and early signals are promising.
For patients whose primary concern is cardiovascular risk alongside weight management, semaglutide’s proven outcome data may tilt the decision. Cardiac history is a standard part of the evaluation at both the Franklin and Nolensville clinics before any GLP-1 prescription.
Semaglutide is available as a once-daily oral tablet, which is currently the only FDA-approved GLP-1 pill for weight management. Tirzepatide is injection-only. For patients who have significant needle anxiety or strongly prefer oral medications, this availability difference can be the deciding factor between the two classes.
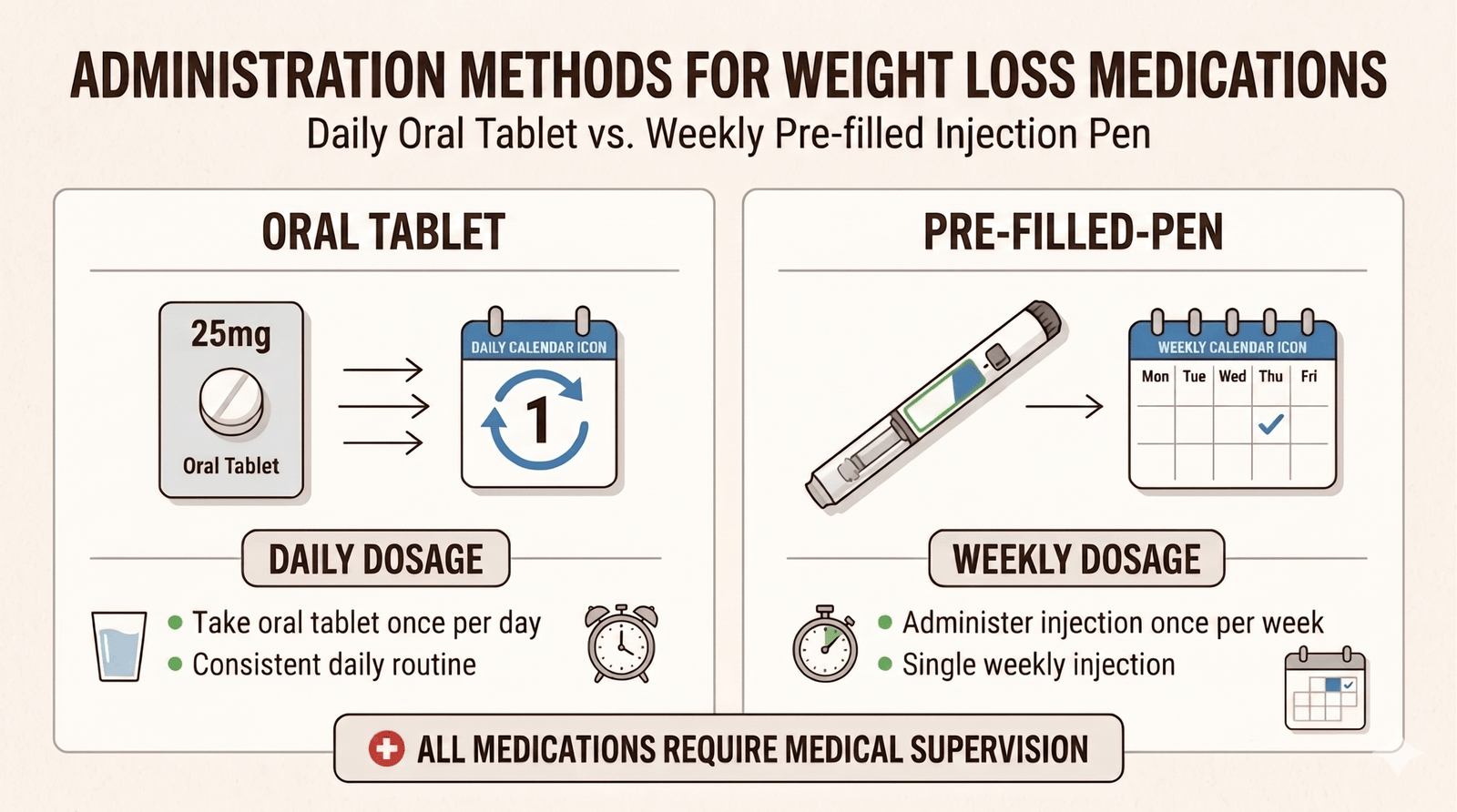
It is worth noting that the injectable forms are dosed once weekly, while the oral tablet is taken daily on an empty stomach with specific water and fasting instructions. Many patients find the weekly injection more convenient once they get past the initial apprehension about self-injection. Either route can work; the question is which fits your lifestyle and comfort level.
The right choice depends on your health history, weight loss goals, and how your body responds. Tirzepatide tends to be the stronger option when maximum weight loss is the priority, when you have significant insulin resistance, or when you have plateaued on semaglutide and need a different approach. The dual mechanism gives it an edge in patients without diabetes in particular.
Semaglutide tends to be the better choice when you have established cardiovascular disease, when you want the longer safety track record, when you prefer or need an oral option, or when you value appetite-focused weight loss support with proven outcome data. Patients who cannot tolerate one medication often tolerate the other better, so switching is always on the table.
Before starting either medication, your provider will review your personal and family history of thyroid cancer, pancreatitis, gallbladder disease, kidney function, and pregnancy plans. Body Works physicians evaluate every patient individually at the Franklin and Nolensville clinics, and adjust treatment as your response dictates. Schedule a Free Consultation to discuss which GLP-1 medication fits your goals.
Medically reviewed by Dr. Donald Vollmer, MD
Managing Physician, Body Works TN

If you have been struggling to lose weight despite trying diet after diet, you are not alone. For many people in Franklin, TN, Nolensville, TN,

Walking into a weight loss clinic for the first time stirs up a specific kind of anxiety. You have already tried diets, gyms, maybe even

Medical weight loss in Franklin and Nolensville, TN combines GLP-1 medications with physician supervision to address the hormonal and metabolic factors that make weight loss

You finish breakfast, and before the last bite is swallowed, your mind is already planning lunch. You walk past a bakery and the smell hijacks

Everyone seems to know someone who dropped 20 pounds in a month on one of the new GLP-1 medications. The before-and-after photos are everywhere. The

Choosing between an in-person and online GLP-1 weight loss program is one of the most consequential decisions a patient can make before starting treatment. The