GLP-1 Weight Loss for Women in Menopause and Perimenopause: What Changes and What Helps
The weight gain that arrives during perimenopause is genuinely different from weight gain at other times of life. It often shows up around the midsection
Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the first-line choice for many physicians. The reason is not marketing. It is mechanism. Tirzepatide activates two metabolic receptor systems simultaneously, where semaglutide activates one, and that single design difference produces measurably larger weight loss in head-to-head clinical comparisons.
For patients researching tirzepatide as a primary option, or considering switching from semaglutide, the questions that matter are not the same as the general GLP-1 questions. Tirzepatide has its own dosing schedule, its own side-effect profile, its own response pattern, and its own clinical fit. Treating it as interchangeable with semaglutide misses the parts that make it the right choice for some patients and the wrong choice for others.
This guide walks through how tirzepatide works, what the SURMOUNT trial data actually shows, the dosing schedule from week one through maintenance, the side-effect picture compared to semaglutide, and what to expect if you start treatment at a physician-supervised program. If you are still deciding between the two medications, our comparison post covers that question more directly. This post goes deeper on tirzepatide as a standalone clinical decision.

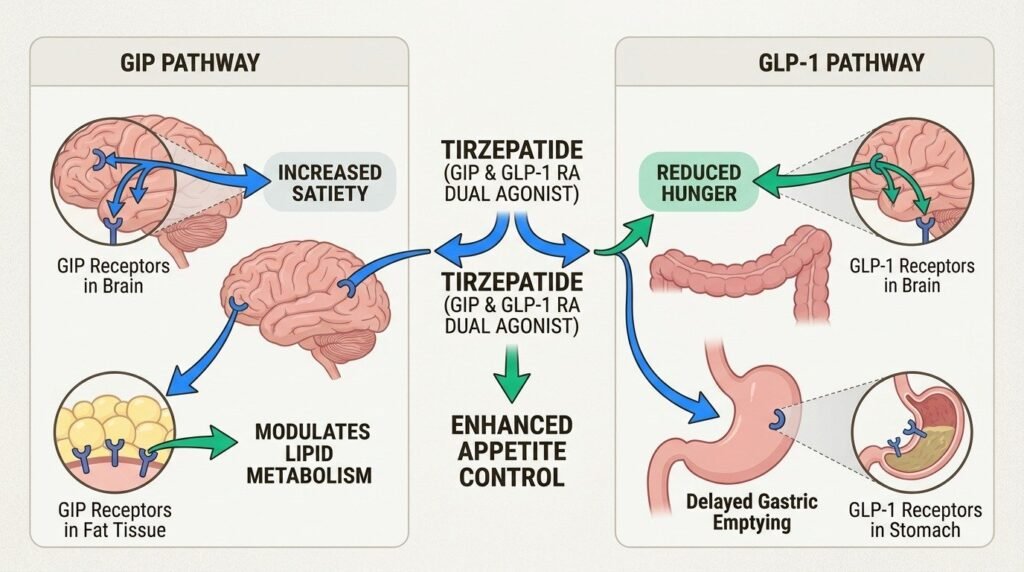
Tirzepatide is a dual agonist that activates both the GLP-1 receptor and the GIP receptor (glucose-dependent insulinotropic polypeptide). Semaglutide activates only the GLP-1 receptor. GIP is a second incretin hormone that, like GLP-1, is released from the gut after eating and influences insulin secretion, satiety, and energy balance.
The dual mechanism is not just additive. The two receptors interact in ways that appear to amplify each other’s effects on appetite suppression and metabolic regulation. Patients on tirzepatide often report stronger and more sustained satiety than they experienced on semaglutide, particularly at the higher dose levels. The slowed gastric emptying that drives GLP-1 nausea is also present with tirzepatide, but the GIP component appears to attenuate it somewhat, contributing to a slightly better tolerability profile in some patients.
The same dual mechanism affects metabolic markers beyond weight. Tirzepatide produces larger improvements in HbA1c, fasting glucose, and lipid markers than semaglutide does, on average. For patients whose weight gain is intertwined with prediabetes, type 2 diabetes, or metabolic syndrome, the dual-agonist effect on glucose handling is clinically meaningful in addition to the weight loss benefit.
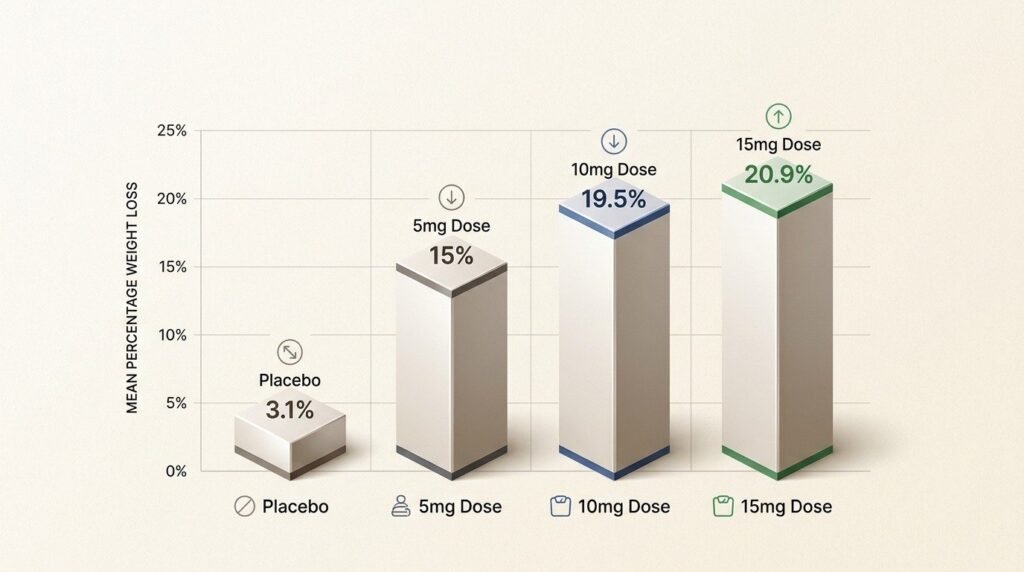
The pivotal trial for tirzepatide in chronic weight management was SURMOUNT-1. Adults with obesity received tirzepatide at 5 mg, 10 mg, or 15 mg weekly, or placebo, for 72 weeks. Mean weight loss at the highest dose was 22.5%, compared to 2.4% on placebo (Jastreboff et al., New England Journal of Medicine, 2022). The 10 mg dose produced 21.4% weight loss, and the 5 mg dose produced 16.0%.
For a 220-pound patient, 22.5% works out to approximately 50 pounds. For comparison, semaglutide 2.4 mg in the STEP 1 trial produced 14.9% weight loss over 68 weeks, which is roughly 33 pounds for the same starting weight. The gap is not subtle. Tirzepatide produces more weight loss on average.
The trial also showed that the share of patients hitting larger weight loss thresholds was substantially higher on tirzepatide. Roughly 40% of patients on tirzepatide 15 mg lost at least 25% of their body weight. About 15% lost at least 30%. Those numbers are well above what semaglutide produces and put tirzepatide in a weight loss range that previously was achievable only with bariatric surgery for many patients.
Subsequent trials extended the picture. SURMOUNT-2 evaluated tirzepatide in adults with type 2 diabetes and obesity, finding 12.8% weight loss at 15 mg over 72 weeks, smaller than the non-diabetic population but still substantially larger than semaglutide produces in the same group (Garvey et al., The Lancet, 2023). SURMOUNT-4 confirmed that continued treatment preserved weight loss and that discontinuation produced regain, in line with the pattern seen with semaglutide.
The dose-by-dose breakdown from SURMOUNT-1 makes the response curve concrete:
| Tirzepatide dose | Mean weight change at 72 weeks | Patients losing 20% or more |
|---|---|---|
| 5 mg | -15.0% | 30.0% |
| 10 mg | -19.5% | 50.1% |
| 15 mg | -20.9% | 56.7% |
Most of the weight-loss benefit is captured by the 10 mg dose. The step from 10 mg to 15 mg adds an additional reduction, but the curve flattens, and the side-effect cost rises. For many patients, 10 mg is the right long-term maintenance dose; the decision to push higher is a clinical judgment that weighs marginal benefit against tolerance.
Tirzepatide is administered as a once-weekly subcutaneous injection. The titration schedule starts at 2.5 mg and steps up at four-week intervals, with a target maintenance dose typically between 5 mg and 15 mg depending on the patient’s response and tolerance.
The standard schedule for chronic weight management:
Not every patient titrates to 15 mg. Some hold at 5 mg or 10 mg as their long-term maintenance dose because they have reached a satisfactory weight or because side effects do not justify pushing higher. The clinical decision at each step is whether the additional appetite suppression and weight loss are worth the additional side effect risk for that patient.
The side-effect profile of tirzepatide is similar in pattern to semaglutide but somewhat different in intensity. Gastrointestinal effects, especially nausea, are the most common reason patients discontinue either medication. The trial data shows nausea in approximately 24 to 33% of tirzepatide patients depending on the dose, compared to 44% on semaglutide 2.4 mg. The difference is real but modest, and individual patient experience varies considerably.
Other GI symptoms, including diarrhea, constipation, and reflux, occur at rates similar to semaglutide. Most are mild, dose-dependent, and improve over the first few weeks at each dose step. Following the standard titration schedule, eating smaller meals, avoiding high-fat foods, and staying well hydrated all help. Patients who push through the titration steps faster than the schedule recommends typically experience more severe GI symptoms (FDA prescribing information for incretin-based therapies).
The non-GI side-effect profile is similar between the two medications. Hair shedding is reported by a small percentage of patients on either drug, with the same telogen effluvium mechanism tied to rapid weight loss. Muscle loss is a concern with both medications and is addressed with the same protein-and-resistance-training protocol. Hypoglycemia is rare in non-diabetic patients but possible in patients on other glucose-lowering medications.
Tirzepatide carries the same boxed warning as semaglutide regarding medullary thyroid carcinoma based on rodent studies. The clinical relevance in humans remains uncertain, but patients with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 are not candidates for either medication.
The candidacy criteria for tirzepatide overlap heavily with semaglutide. The standard threshold is a BMI of 30 or higher, or a BMI of 27 or higher with weight-related comorbidities. The same contraindications apply. The same lab and history workup is appropriate before starting.
Specific patient profiles that often favor tirzepatide over semaglutide include patients with substantial weight loss goals where the additional 5 to 10 percentage points of expected loss matters; patients with co-existing type 2 diabetes or significant insulin resistance, where the dual-receptor mechanism produces larger improvements in glucose handling; and patients who have plateaued on semaglutide and want to access additional weight loss through a different mechanism. Our broader candidacy guide covers the foundational eligibility criteria that apply to both medications.
Patients who tolerated semaglutide poorly may or may not tolerate tirzepatide better. The mechanism overlap means that a patient with severe nausea on semaglutide is more likely than average to have nausea on tirzepatide too, though the rates are somewhat lower. The decision to switch is a clinical judgment that should weigh tolerance history alongside expected response.
The initial consultation for tirzepatide treatment at Body Works covers the same ground as a semaglutide consultation: comprehensive lab work, a detailed health history, body composition assessment, and a discussion of goals and lifestyle context. The medication choice is made within that broader picture rather than as a default first step.
For patients new to GLP-1 treatment, the conversation about whether to start with semaglutide or tirzepatide considers the weight loss goal, the metabolic profile, the side-effect tolerance history with other medications, and cost considerations. For patients switching from semaglutide, the conversation includes why the switch is being considered, what response was seen on semaglutide, and what dosing transition makes sense.
The titration schedule, follow-up cadence, and the supporting infrastructure for protein, resistance training, and lab monitoring are all built into the plan from the start. Patients leave the initial consultation with a clear protocol, not a vague prescription. Schedule a free consultation to talk through whether tirzepatide is the right starting point for your situation, or whether a different medication or sequence makes more clinical sense.
Medically reviewed by Dr. Donald Vollmer, MD
Managing Physician, Body Works TN

The weight gain that arrives during perimenopause is genuinely different from weight gain at other times of life. It often shows up around the midsection

For most of 2023 and 2024, semaglutide was on the FDA shortage list. That single regulatory designation opened the door for compounding pharmacies to legally

The most common question patients ask after their first injection is some version of “when will I see results?” That question is reasonable, but the
Hair shedding ranks consistently among the top concerns patients raise before starting semaglutide or tirzepatide. Search volume on “does semaglutide cause hair loss” has climbed
Most patients on semaglutide or tirzepatide eventually ask the same two questions: how long do I need to take this, and what happens if I

If you have been struggling to lose weight despite trying diet after diet, you are not alone. For many people in Franklin, TN, Nolensville, TN,