
Tirzepatide Deep Dive: Dosing, Results, and What Patients Should Expect
Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
For most of 2023 and 2024, semaglutide was on the FDA shortage list. That single regulatory designation opened the door for compounding pharmacies to legally produce semaglutide for patients who could not access the brand-name version. By the time the shortage ended, hundreds of thousands of patients were receiving compounded semaglutide through telehealth platforms, weight loss clinics, and direct-to-consumer pharmacy channels. The supply pattern reshaped the entire weight loss medication market.
The shortage status changed in 2025, and the rules around compounding tightened. Some compounded semaglutide is still legally produced under specific clinical conditions. Some is not. The difference matters because not every vial labeled “compounded semaglutide” is the same product, and the variation between sources includes safety-relevant details that patients usually do not see on the label.
This guide explains what compounded semaglutide actually is, where the safety lines fall, and what to ask any provider, telehealth service, or pharmacy before you take a vial home. The goal is not to argue that compounded semaglutide is universally bad or universally fine. The goal is to give you the questions that separate a careful provider from a careless one, because that distinction is what determines whether the medication you are taking is what you think it is.

Compounded medications are drugs prepared by a licensed pharmacy from raw active pharmaceutical ingredients, rather than dispensed from a manufacturer-finished sealed vial. Compounding is a legitimate, long-established part of pharmacy practice. It exists to handle clinical situations where a manufactured product does not fit a specific patient’s needs, such as a patient who is allergic to a preservative in the manufactured version, a child who needs a smaller dose than the manufactured strength, or a patient whose medication is in legitimate shortage.
The Federal Food, Drug, and Cosmetic Act allows compounding under two distinct categories. Section 503A pharmacies are state-licensed and produce custom preparations for individual patients with valid prescriptions. Section 503B outsourcing facilities are federally registered and can produce larger batches under stricter quality standards, often supplying clinics directly. Both categories were active producers of compounded semaglutide during the shortage.
The active pharmaceutical ingredient itself, semaglutide, is the same molecule whether it comes from a brand-name vial or a compounded preparation. What differs is the supply chain for the raw material, the formulation excipients, the quality of the compounding process, the sterility and potency testing, and the regulatory oversight applied to each lot.
503A pharmacies are regulated primarily at the state level. They must operate under a valid prescription for an individual patient, and the compounding process is reviewed by state boards of pharmacy. Quality varies, sometimes significantly, depending on the specific pharmacy. Some 503A pharmacies operate to standards that approach 503B levels. Others do not.
503B outsourcing facilities are registered with the FDA, inspected against current Good Manufacturing Practices, and required to meet stricter standards for testing, documentation, and recall procedures. The compromise is that 503B preparations cannot be dispensed for an individual patient on a one-off basis the way 503A preparations can. They are typically supplied in batches to clinics for in-office administration.
For a patient evaluating a compounded semaglutide source, the first practical question is which type of pharmacy supplies it. The second is whether that pharmacy publishes inspection records and test results. A reputable supplier provides this information without resistance. A supplier that cannot or will not is a signal that the patient should look elsewhere (National Association of Boards of Pharmacy).

The brand-name versions of semaglutide are formulated as the semaglutide base. Some compounded preparations during the shortage period were produced using semaglutide salt forms, specifically semaglutide sodium and semaglutide acetate. The FDA issued a compounding alert in 2023 noting that these salt forms are not the same active ingredient as approved semaglutide and have not been demonstrated to be safe and effective in humans (FDA Drug Safety Communication on compounded semaglutide).
The salt-form issue matters because two preparations labeled “semaglutide” are not necessarily delivering the same molecule to your bloodstream. A patient who tolerates and responds well to semaglutide base may have a different experience with a salt-form preparation, and the safety data for salt forms in chronic dosing is essentially nonexistent.
A patient considering a compounded source can simply ask: “Is this compounded from semaglutide base, or from a salt form?” A provider who does not know the answer is not in a position to advise you on the safety of the medication they are prescribing. A provider who specifies semaglutide base and can document the source of the active ingredient is operating at a different level.
When semaglutide was on the FDA shortage list, compounding under section 503A was broadly permitted as a way to maintain patient access. The shortage status changed in 2025, and the rules tightened. Compounded semaglutide is still legally produced for patients with documented clinical reasons that the manufactured product does not fit, but the bar for what counts as a valid clinical reason is higher than it was during the shortage.
Some telehealth services that scaled rapidly during the shortage are now operating in a more constrained environment, and the legality of certain mass-market compounded prescriptions is in active dispute. Patients who started compounded semaglutide during the shortage and are still on it should understand that the clinical and regulatory landscape has shifted, and that a careful provider review is appropriate.
This is one of several reasons that choosing between in-person and online GLP-1 care is a more consequential decision than most patients realize. An in-person provider can re-evaluate the source, the dose, and the appropriateness of the medication as the regulatory environment changes. A subscription telehealth platform that ships a vial every month may not perform that re-evaluation at all.
Several specific questions reveal a lot about a compounded supplier’s quality. Asking them, and noticing how they are answered, separates the providers worth working with from those worth avoiding.
Is the pharmacy a 503A or 503B facility, and can the inspection records be shared? Both types can produce safe preparations, but the patient deserves to know which they are receiving and to verify the pharmacy’s regulatory standing.
Is the active ingredient semaglutide base, and where is it sourced? Semaglutide base is the form used in the manufactured products and is what the long-term safety data supports. Salt forms are a different molecule and lack that data.
What are the sterility and potency test results for the lot being dispensed? Reputable compounding pharmacies test each lot for sterility, endotoxin, potency, and stability. The results are documentable. A pharmacy that cannot produce them on request is not operating at the standard a patient should accept.
Who reviews adverse events, and what is the process for reporting them? Patients on any compounded medication should have a defined channel for reporting side effects, and the provider should be tracking and aggregating the reports. Subscription services that disclaim responsibility for adverse events at the click-through level are not the right place to take a chronic medication.
The same checklist applies to any prescription a patient receives, but it is especially important for compounded preparations where the regulatory backstop is thinner than for manufactured products. Our guide to spotting weight loss medication scams covers the broader red flags, and most of them apply to evaluating compounded sources as well.

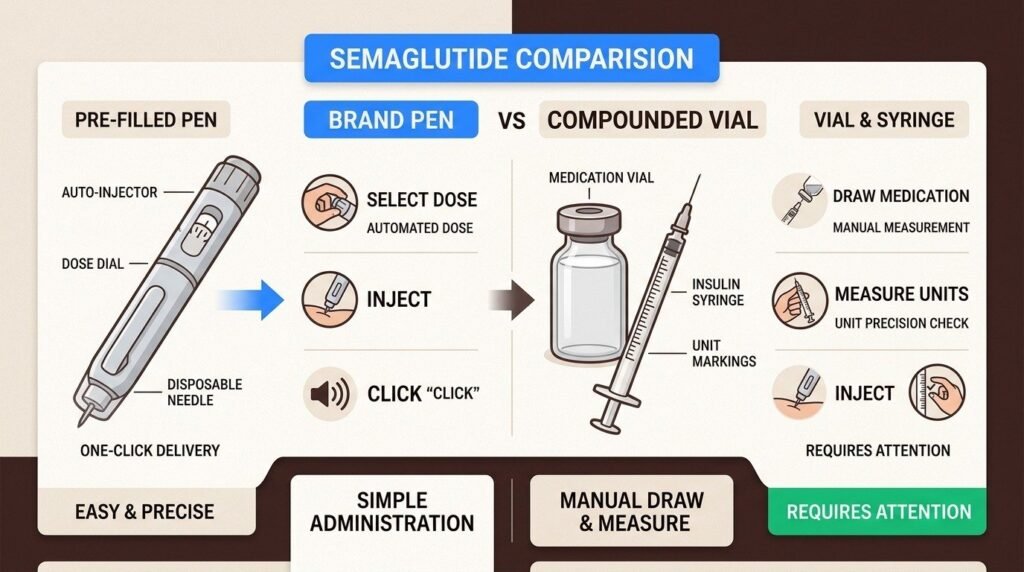
Body Works does not subscribe to a single sourcing model that fits every patient. The right product, dose, and supply chain depend on the patient’s clinical profile, response history, and the current regulatory context. What is consistent is the diligence applied to whichever source is used. Pharmacy partners are vetted for licensure, inspection history, and willingness to share testing documentation. The active ingredient is verified as semaglutide base when compounded preparations are appropriate. The dose, formulation, and supply are reviewed at each follow-up visit, not assumed to be unchanged from the prior month.
Patients are also informed about what they are being prescribed, why, and what the alternatives are. If a brand-name product is the right fit, that is the recommendation. If a compounded preparation from a vetted 503B facility is the right fit, that is the recommendation, and the reasoning is documented. The point is that the decision is clinical, not commercial.
The patients most at risk from compounded semaglutide are not the ones receiving it through a careful program with documented sourcing. They are the ones receiving it through high-volume subscription channels with minimal oversight, where the product, the active ingredient, and the dose may not match what the label claims. Schedule a free consultation if you are currently on a compounded preparation and want a second opinion on the source and the protocol you are following.
Medically reviewed by Dr. Donald Vollmer, MD
Managing Physician, Body Works TN

Tirzepatide is the newer of the two leading weight loss medications, and over the past two years it has been quietly displacing semaglutide as the
The weight gain that arrives during perimenopause is genuinely different from weight gain at other times of life. It often shows up around the midsection

The most common question patients ask after their first injection is some version of “when will I see results?” That question is reasonable, but the
Hair shedding ranks consistently among the top concerns patients raise before starting semaglutide or tirzepatide. Search volume on “does semaglutide cause hair loss” has climbed
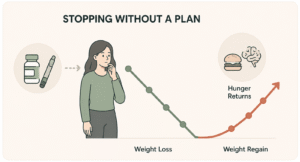
Most patients on semaglutide or tirzepatide eventually ask the same two questions: how long do I need to take this, and what happens if I

If you have been struggling to lose weight despite trying diet after diet, you are not alone. For many people in Franklin, TN, Nolensville, TN,